3-part, 4-part or 5-part veterinary hematology analyzers: advantages and disadvantages
Automated hematology analyzers are frequently used in veterinary clinics and settings to assess and monitor the health condition of patients. Although 3-part hematology analyzers provide sufficient information for most clinical settings, trends show an increased interest in 5-part instruments.
While 5-part analyzers sometimes can offer improved assessment of white blood cells, 3-part instruments offer great cost benefits. Here, we describe clinical settings, where 3-part differentials and 5-part differentials can provide different advantages.
Introduction
A complete blood count (CBC) is the most requested analysis by veterinarians to assess and monitor patients’ health condition. For this, automated hematology analyzers are frequently used in clinical laboratories to measure the oxygen-carrying red blood cells (RBC), the platelets (PLT) that help clot the blood, and the white blood cells (WBC) of the immune system. As part of the CBC, a differentiation of the WBCs into their subgroups is typically conducted. In normal mammalian blood, these subgroups are usually divided into five major categories:
• The neutrophils (NEU) account for about 60% of the WBCs. NEUs are the first line of defense and help fight mainly bacteria (and fungi) through phagocytosis, and a high count (> 85%) can therefore indicate a bacterial infection.
• Lymphocytes (LYM), accounting for about 30% of all WBC, are part of the adaptive immune system and mainly help fight viruses. A high LYM count can therefore be an indication of a viral infection. On the other hand, a low LYM count with a slight MONO elevation is a common finding in especially cats due to stress.
The last 10% comprises of:
• Monocytes (MONO)
• Eosinophils (EOS)
• Basophils (BASO)
The EOS and BASO are typically associated with allergies or parasite infections and the BASO tend to constitute of < 1% in animals.
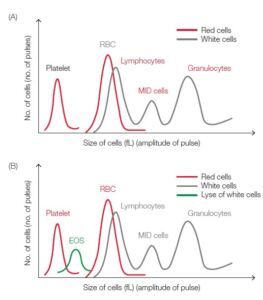
Fig 1. Test result from (A) 3-part and (B) 4-part hematology analyzer visualized in a histogram.
A high MONO count, for example, can indicate a chronic inflammatory disease, whereas high EOS counts give an indication of asthma, an allergic reaction, or a parasite infection. A high BASO count is typically associated with inflammatory reactions, especially those causing allergic symptoms, as well as heartworm and neoplasia together with high levels of eosinophils. High numbers of the WBCs can also be an indication of certain forms of cancers, such as leukemia or lymphoma.
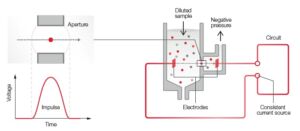
Fig 2. The principle for measuring changes in the electrical impedance produced by a cell passing through an aperture.
Automated hematology analyzers
In today’s general screenings, CBC tests are performed using automated hematology analyzers. In addition to reporting RBC, PLT, and WCB counts, an analyzer also measures the oxygencontaining hemoglobin (HGB) and determines a range of other parameters such as the mean cell volume (MCV), PLT width distribution (PDW), and hematocrit (HCT), that is, the red blood cell-to-plasma ratio. Hence, an automated analyzer can provide much more information than a manual count in some cases.
A 3-part instrument commonly uses impedance to differentiate WBC into granulocytes (mainly neutrophils, but also eosinophils and basophils), lymphocytes, and MID cells (mainly monocytes, but also eosinophils) based on cell size (Fig 1). Each cell passing through the aperture causes a drop in the electrical current (a pulse). The number of generated pulses correlates with the number of cells, whereas the size of the pulse is related to the cell size (Fig 2). In addition to impedance, a 5-part instrument employs the
principle of flow cytometry to differentiate WBC into their five major sub-populations—neutrophils, lymphocytes, monocytes, eosinophils, and basophils—based on cell size and complexity (granularity) (Fig 3). In flow cytometry, cells are forced to flow in a single file through the aperture by a sheath fluid, created
by a fast-moving diluent that surrounds the slow-moving sample (Fig 4A). A laser beam is passed through the sample, and when a cell passes through the sensing zone, the light is scattered and measured by a photoconductor that converts the light into an electrical impulse. The number of generated impulses
correlates with the number of cells, whereas the light scatter is used to determine cell granularity, shape, and size (Fig 4B).
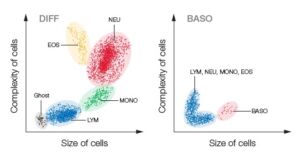
Fig 3. Test results from a 5-part hematology analyzer are visualized in a 4-part differential scattergram and a separate scattergram for BASO. Ghost = nucleated RBC, lyse-resistant RBC, and platelet clumps.
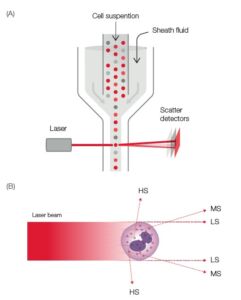
Fig 4. (A) Laser-based flow cytometry for 5-part differential of white blood cells. (B) Three-angle laser-scatter method, where the low angle signal (about 1° to 5°) represents the cell volume information, the middle angle signal (about 7° to 20°) represents the cell nucleus information, and the high angle signal (about 90°) represents the cell nucleus and cytoplasm information.
3/4-part versus 5-part analyzers
Although each WBC sub-type provides information that helps diagnose blood-related conditions, a 3-part instrument will provide sufficient information required for most clinics. With a simple CBC, the neutrophil and lymphocyte counts will answer the question of a viral infection or a bacterial infection that can
be treated with antibiotics (Fig 5). On the veterinary side there are also 4-part analyzers on the
market which allow for an eosinophil differentiation, giving additional information of for instance parasitic infections and allergies (1). For specialty laboratories, a 4-part or a 5-part instrument can therefore provide a more detailed and targeted assessment of the blood status, for example, to distinguish eosinophils and basophils from neutrophils (Fig 6A, B, C, D and E). Currently, studies on the veterinary market seem to
conclude that no automated 5-part analyzer can give a good enough correlation for basophils, therefore they should only be checked under microscope (2). Most likely one reason is the difficulty due to the inter-species variations for eosinophil and basophil morphologies, making it difficult for one analyzer to be able to distinguish between these cell subtypes for several different animal species using the same reagents, etc. (3).

Fig 5. Histograms from (A) a normal sample, (B) a sample from patient with a virus infection, and (C) a sample from patient with a bacterial infection.
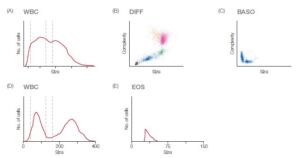
Fig 6. (A) Abnormal 3-part histogram, where lymphocytes cannot be differentiated from MID cells, and MID cells not from granulocytes. (B, C) Scattergrams from a 5-part instrument, showing a 4-part differential with a clearly separated eosinophil population, and the basophile population displayed in a separate diagram. (D) Histogram from a 4-part analyzer showing lymphocytes, neutrophils and monocytes separated and (E) an eosinophil histogram separately showing the eosinophil population clearly identified.
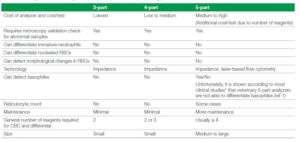
Table 1. advantages and disadvantages of 3-, 4-, and 5-part veterinary hematology analyzers
One advantage of certain 5-part laser analyzers can be for instance 5-part instruments with optical PLT, which seem to slightly reduce the feline platelet aggregation issues. However, this larger, expensive analyzer is usually situated in reference and university animal hospitals and not in general clinic settings due to cost, size and complexity.
Another potential advantage of 5-part analyzers is reticulocyte count in the diagnosis of regenerative versus non-regenerative anemias. However, reticulocytes do not give you the full anemia overview and the RBC morphology needs to be checked under the microscope. Moreover, cats have punctuate and aggregate reticulocytes which need to be differentiated manually. To read more about anemia (4). Depending on local policies, samples that are flagged as abnormal commonly require microscopical examination to confirm the results obtained with the analyzer, regardless of instrument type. For this, blood smears are either examined in the clinic or typically sent to a reference laboratory for a manual count.
For general screenings, a 3-part instrument can contribute to an improved laboratory economy by offering the caregiver a greater profit opportunity. In addition, a 3-part instrument based on robust impedance technology might require less maintenance than a 5-part instrument, which also includes a more sensitive
and complex laser-based measurement technology.
Exigo™ H400
Exigo H400 is designed with the patient and end-user in focus to be a cost-efficient, user friendly and accurate instrument. Exceptionally, Exigo H400 veterinary hematology analyzer together with reagents, controls and calibrators are all manufactured by one company, Boule, which gives a total traceability and quality concept.
All hematology analyzers on the market today are screening tools, with a purpose to identify abnormal samples and minimize manual microscopy of healthy individuals, saving cost and time.
To achieve this, the most important is to have an accurate, reliable analyzer which is cost efficient and gives you clinically relevant parameters. Measurement technology and design With a closed shear-valve and a volumetric based counting, the Exigo H400 is unique among other impedance analyzers (5), it allows for Exigo H400 to have one of the lowest coefficient of variations (CV) for the CBC parameters on the
market, see Table 2.
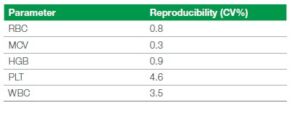
Table 2. Reproducibility, measured as an average of 10 measurements each on 9 different dog venous blood in K2-EDTA collected normal samples, on 3 instruments, in open tube mode
4-part
The H400 is a 4-part analyzer, meaning it provides NEU, LYM, MONO and EOS differentials to the end-user. Exigo H400 uses a reagent-based eosinophil counting, meaning there is a separate lyse used only for the eosinophil counting. This is to ensure that all other WBC are lysed apart from the EOS remaining (5).
Cost efficiency With the possibility to choose between a 3-part or a 4-part analysis Exigo H400 also becomes very cost efficient as only two reagents are required for a 3-part analysis and three reagents for a 4-part sample analysis.
Low maintenance
Designed with few moving parts, a maintenance-free closed shear valve design, and with the majority of the instrument cleaning procedures being automated, the user maintenance of Exigo H400 analyzer is kept to a minimum. To minimize user intervention, the analyzer performs automatic cleaning every 48 hours, using diluent. Moreover, on the Exigo H400 system, there is an additional cleaner on-board which is used during an enzymatic cleaning cycle to dissolve proteins and for clot prevention each time after exit standby.
Additionally, an automatic clot prevention is also performed every 24 hours. All these automatic maintenance steps ensure the robustness of the Exigo H400 which also allow for the user to run body
fluids that otherwise would cause protein and clots.
Clinical help
For abnormal samples the Exigo H400 analyzer will present the user with flags. These come as either instrumental flags or pathological sample messages. The pathological sample messages are to help guide the user to potential next steps in the identification of the underlying diagnosis of the patient.
For example, one pathology message could be a high MCV value together with the message “MCV: Evaluate histogram; RBC morphology on slide.” Among these messages are also warnings such as hemolysis and heinz-bodies.
Conclusion
For most cases, the same decisions can be made with a 3-part hematology analyzer as with a 5-part analyzer. Typically, a 3-part instrument is also quicker and cheaper. If abnormal values are obtained, microscopy is still required, irrespective of instrument type. Although 3-part differentials provide excellent precision for general screenings, accuracy is improved with 5-part differentials for abnormal samples, reducing the number of manual blood smears. Regardless of the choice of a a 3-part, 4-part or a 5-part instrument, of utmost importance is the ability of the analyzer to detect and flag for abnormal samples. With Exigo H400,
you also have the additional benefits of having an eosinophil count though still with a cheaper, less maintenance, robust and quicker impedance analyzer.
References
1. Application note: Importance of eosinophil parameter on veterinary hematology analyzers. Boule Diagnostics, ANE34070, Edition 1 (2019).
2. Errors in basophil enumeration with 3 veterinary hematology systems and observations on occurrence of basophils in dogs, Lilliehöök I., Tvedten H.W., Veterinary Clinical Pathology ISSN 0275-6382, DOI:10.1111/j.1939-165X.2011.00353.x, (2011).
3. White Paper: Introduction of veterinary hematology parameters: factors affecting parameters, clinical relevance and inter-species variations. Boule Diagnostics, WP34071, Edition 2 (2019).
4. White Paper: Veterinary classifications and investigation of non-regenerative and regenerative anemia and the role and clinical relevance of reticulocytes. Boule Diagnostics, WPE35331, Edition 1 (2020).
5. Tech note: Exigo H400 analyzer design for optimized system performance. Boule Diagnostics, TNE33601,
Edition 1 (2019).

Fig 4. User interface of Exigo H400 with a pathological sample message through the information button “i”.
PDF download
White paper: White paper 3p-or-5p Veterinary hematology analyzers wpe-35611-2
