Introduction of veterinary hematology parameters: factors affecting parameters, clinical relevance, and inter-species variations
Hematology is the most common diagnostic testing performed, tightly followed by clinical chemistry. To understand the results of a complete blood count (CBC), it is important to understand the meaning of each parameter, how the various cells are produced and where, what factors that may affect their concentrations and production. Veterinary blood is also more complicated than human diagnostics in that species have varying morphologies and factors affecting them.
All blood cells (white blood cells, red blood cells and platelets) are produced in the bone marrow from the pluripotent stem cell. They then divide into various cell lineages and continue their maturation processes before being released into the blood stream.
Boule’s Exigo™ H400 is an automated hematology system designed to be a robust, easy to use and high-quality instrument and prides itself in its extremely high precision and accuracy (1). The analyzer is especially tailored for clinics with small animals, dehydrated animals or body fluid, with patented special features.
Red blood cells and associated hematological parameters
The stem cells differentiate into all the blood cells including the platelets and white blood cells as well as the most abundant blood cell, the red blood cells (RBC).
Where do they come from?
The bone marrow is the source of the production of the RBCs, also known as erythrocytes. The process of differentiating the stem cell to a mature red blood cell, also known as
erythropoiesis, takes slightly different time in different species, e.g. in cows this process takes around 100 hours. Once the RBCs are mature and released into the blood stream, their lifetime is not very long; approximately 143 days in horses, 110 days in dogs, 73 days in cats, and 43 days in mice (2).
During erythropoiesis, the cell nucleus is gradually replaced by hemoglobin (HGB), giving the RBCs their characteristic biconcave shaped discs in dogs and slightly more rounded shape in cat and horses.
It is mainly through a hormone erythropoietin (EPO), produced by the kidneys, that the bone marrow is stimulated to produce more RBCs. When there is low oxygen detected in the blood (hypoxia), which can occur after a bleeding or at high altitudes, the kidneys respond by producing EPO.
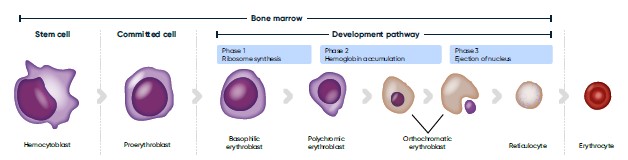
Fig 1. Erythropoiesis and the various maturation stages of the RBCs.
What is their function?
The RBCs have their main task to deliver oxygen from the lungs to the body tissues. This is possible due to the HGB present within the RBCs, which is built up of iron, porphyrin, globulin, etc., and its ability to bind the oxygen. The RBCs along with the HGB also indirectly remove carbon dioxide (CO2), from the body’s tissues back to the lungs, to be exhaled.
How are they measured?
The main RBC parameters:
• RBC concentration
• Mean cell volume (MCV), being an average of the diameter of the RBCs
• Hematocrit (HCT), the packed volume of the red blood cells
• HGB
In “normal” patients, these parameters should all follow each other, whereas in anemic animals these parameters need to be investigated in more depth (3).
Red blood cell concentration (RBC)
The RBC count is one of the fundamental ones also used in the calculations of several other red blood cell indices. Earlier, RBC was calculated in the microscope manually through a Bürker chamber. However, with automated systems the methodological error has decreased from around 5%–10% to less than 1%. It is however important that the automated systems are adapted for the various animal profiles to be tested, as the size of the RBCs vary greatly.
Exigo H400 is especially adapted with software settings to work for different animal profiles with varying RBC size and membranes. Moreover, the system is designed with a 60 μm capillary to be extra sensitive to small RBCs (1).
Hemoglobin (HGB)
The HGB concentration is one of the most common parameters requested by the laboratory, with the main reasons being to monitor RBC changes, liquid balance, and anemia. HGB is a robust parameter, which is not easily affected by handling procedures including storage and transportation. This makes it very reliable.
Exigo H400 utilizes a non-toxic lysing reagent, which allows for a stable HGB complex that is measured through absorption spectrophotometry at a wavelength of 535 nm and compared with a blank for each sample (1).
Hematocrit (HCT)
The HCT is the amount of RBC volume that is present in the total blood volume as a percentage, meaning the packed cell volume of RBCs. It is also an important parameter in anemia where it is used to set the severity of the anemia, polycythemia, and dehydration. The HCT can be measured either by an automated hematology counter (as RBC × MCV) or if an EDTA blood tube is centrifuged, the RBC-whole blood volume can also be measured. These two methods give slightly different results.
Reticulocytes (RETICS)
Figure 1 shows the maturation stages of the RBCs. One of the latest stages is the reticulocytes. Morphology wise they are larger than the mature RBCs and have a nucleus containing RNA that can be stained blue. In dogs, RETICS take around 1–2 days to mature.
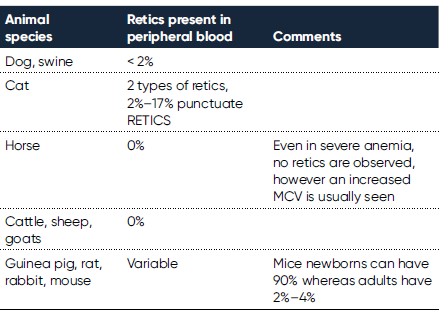
Table 1. Different species and their normal amounts of reticulocytes in the peripheral blood.
Mean cell volume (MCV)
The MCV is also an important indicator for anemia. Being the mean volume of the RBCs, it gives information on microcytosis (small RBCs), macrocytosis (large RBCs), or normocytic cell distribution. Due to the large variation of MCV among species, it is important that the automated cell counter takes various species into account. Exigo H400 accounts for this.
Red cell distribution width (RDW)
The RDW is a value giving you the variation of the RBC size. In Exigo H400, the RDW is calculated from the RBC size distribution curve and is only presented if the MCV value is displayed.
Mean cell hemoglobin concentration (MCHC)
The MCHC is the average concentration of HGB in the total RBC mass. This means it is calculated as: MCHC = HGB/HCT or MCHC = HGB/(RBC × MCV).
The MCHC can also be used in anemia cases to classify them into normochromic (normal HGB concentration in the RBCs) or hypochromic (when the MCHC is low).
MCHC generally does not vary very much and is a good parameter for checking the stability of an automated hematology analyzer. Daily mean MCHC can be used to check the system over a period of time.
Mean cell hemoglobin (MCH)
The MCH is a clinically less frequently used parameter and is the mean concentration of HGB in the RBCs and calculated as MCH = HGB/RBC. Usually the changes in MCH go hand in hand with observed changes in the MCHC.
Differences among species?
There is a fundamental difference in the RBCs of birds, reptiles, and fish compared with other species in that the RBCs still have their nuclei intact. This entails that the RBCs cannot be lysed by the reagent used with a normal automated hematology analyzer, and hence most automated cell counters cannot analyze blood from these animals. The reason for this is that the most common technology used for counting the white blood cells is to utilize them being the only blood population with nuclei and lysing away all the RBCs. For fish, reptiles, and birds, this means that the un-lysed RBCs could be interfering with the total WBC count.
When it comes to camels, llamas, alpacas, and related species, they also have some differences in their RBCs. Their RBC morphology is ellipsoid and the cells are a lot more resilient to lysing. This as well as their shape may also pose problems for automated cell counters, potentially disturbing the MCV measurement which then directly has an impact on HCT and red blood cell distribution width (RDW).
Exigo H400 has solved the challenges of the camel and related species´ RBC counts by allowing for a longer lysing time to allow for the RBCs to properly lyse. Moreover, there is an MCV compensation and other software features that help ensure the MCV is accurate. As for the birds, reptiles, and fish, the RBCs can still be measured in Exigo H400, though some other parameters will be compromised.
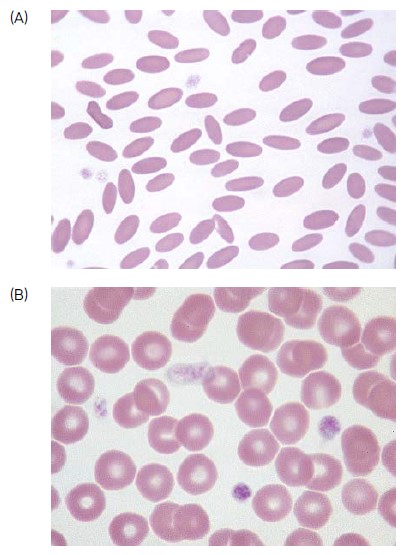
Fig 2. (A) Blood smear from Llama showing the elliptical shaped RBCscompared to (B) a dog smear (4).
Alterations in the red blood cells and associated hematological parameters
The most common clinical diagnosis associated with the red blood cells and the including parameters HGB, MCV, HCT, MCHC, RDW is anemia. Anemia is defined as a too low oxygen carriage capacity of the RBCs. Anemia can be caused by different factors and be of different kind. Anemia will be discussed in more detail further on in this section.
Alterations in RBC, HGB or HCT for clinical relevance
1. Low HGB, HCT or RBC might indicate (2):
• Anemia – can have many causes such as bone marrow failure, EPO deficiency, hemolysis, malnutrition, blood loss, etc.
• Late pregnancy
• Shock – through swelling of the spleen and hypotension
• Anesthesia/sedation – the spleen is more blood-filled
• Age – in younger animals, these parameters tend to be lower, and then increase with age
However, in some cases the results of low HCT can be false.
Potential reasons could include:
• Poor mixing of sample
• Hemolysis
• Low blood volume sampled in EDTA tube in relation to the concentration of EDTA
2. High HGB, HCT, or RBC
With a high concentration of RBC and elevated levels of HCT and HGB, the blood becomes “thicker” and it is less effective to transport oxygen.
Reasons might include:
• Activity, stress, fear – as a response the spleen may contract and release RBCs into the blood. This is especially apparent in warm-blooded horses where the HCT can increase from 35% to 50%. The reason is due to the large reservoir of 1/3 of the blood cells being stored in the resting state of the horse (1).
• Kidney disease – with high EPO
• Dehydration – such as diarrhea
• Anabolic steroid treatment
• Primary or secondary polycythemia – could be caused by high altitude, heart shunt, Cushing’s disease, etc.
However, in some cases, the results of high HGB may be false.
Potential reasons could include:
• Lipemia
• Heinz bodies
• Extended sample storage – causes the RBCs to swell and
the MCV and HCT increase
• High WBC count
The Exigo H400 compensates or high WBC counts using a compensation factor.
PDF download
White paper: White paper parameters and species Exigo H400_WPE_34071-6
