Chemotherapy-induced thrombocytopenia (CIT) is a common complication in cancer patient undergoing treatment. As platelets (PLTs) help the blood to clot, a low count (thrombocytopenia) may cause excessive bleeding in case of wounding. According to literature, a normal reference interval for PLTs in adults is about 160–390 × 109/L (1).
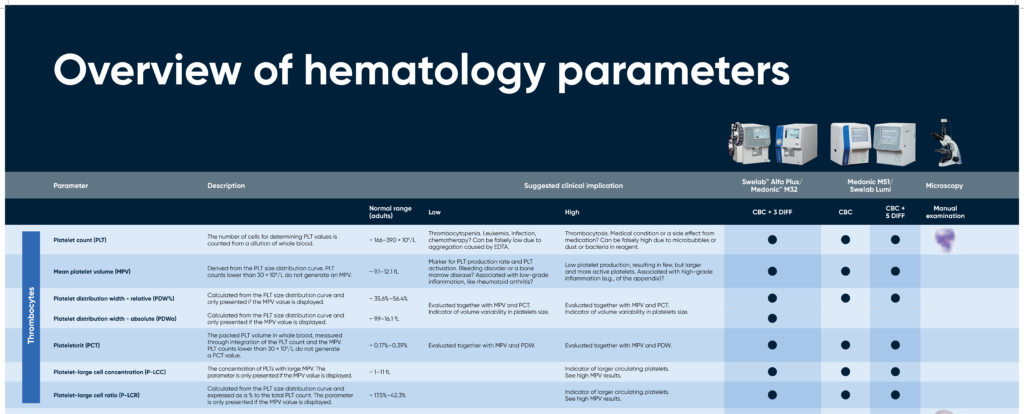
Overview of hematology parameters
Kuter and colleagues at Harvard Medical School, Boston, MA, USA have defined CIT as a platelet count below 100 × 109/L, divided into the following grades (2):
• Grade 1: 75 × 109/L to < 100 × 109/L
• Grade 2: 50 × 109/L to < 75 × 109/L
• Grade 3: 25 × 109/L to < 50 × 109/L
• Grade 4: less than 25 × 109/L
PLT counts below 50 × 109/L can complicate surgical procedures, whereas spontaneous bleeding may occur at a PLT count below 10 × 109/L (2). To predict the bleeding risk, cancer patients receiving chemotherapy is therefore often regularly checked for PLT count.
To accurately determine PLT count in the critically low range, Boule hematology analyzers feature PLT extended count. When enabled, the counting time is extended three times, counting three times as many platelets for a more reliable reporting of the PLT parameter value.
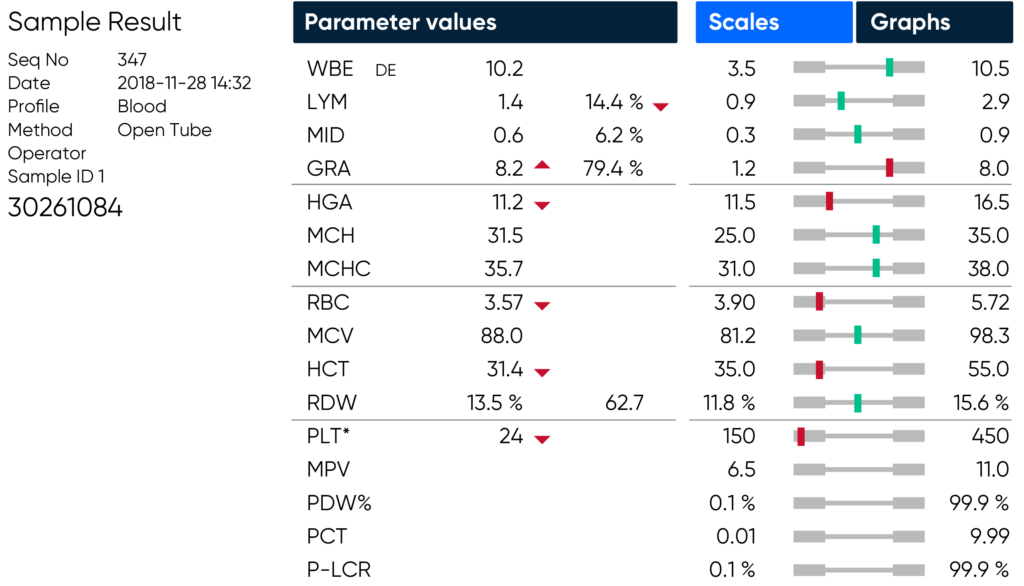
If PLT extended counting time is enabled and a low PLT is detected during analysis, the extended counting time will be displayed on the counting phase screen, and then indicated by an asterisk (*) adjacent to the PLT parameter on the result screen and in printouts and exported PDFs.
Learn more
Learn more about how the design of Boule’s products and solutions may support clinical decision-making .
Reference
1. Mayo Clinic Laboratories. Rochester Interpretive Handbook (2021).
2. Kuter, D.J. Treatment of chemotherapy-induced thrombocytopenia in patients with non-hematologic malignancies. Haematologica 107, 1243–1263 (2022).




